The throttling effect is to reduce the inlet pressure by a constant enthalpy process (represented by horizontal line AB). The polytropic process equation can describe multiple expansion and compression processes which include heat transfer. What do u mean by polytropic process?Ī polytropic process is a thermodynamic process that obeys the relation: where p is the pressure, V is volume, n is the polytropic index, and C is a constant. No, all thermodynamic processes are not polytropic. What is M in polytropic process?įor polytropic process pVn= constant, Cm ( molar heat capacity) of an ideal gas is given by: … B.Cv,m+R(1−n) Are all processes Polytropic? If system volume contracts under a force, work is done on the system. If system volume expands against a force, work is done by the system. Some examples are vapors and perfect gases in many non-flow processes, such as: n=0, results in P=constant i.e. Polytropic processes are internally reversible. Polytropic process is one which obeys the law given below throughout the process. Isentropic process is one in which the entropy remains constant throughout. 
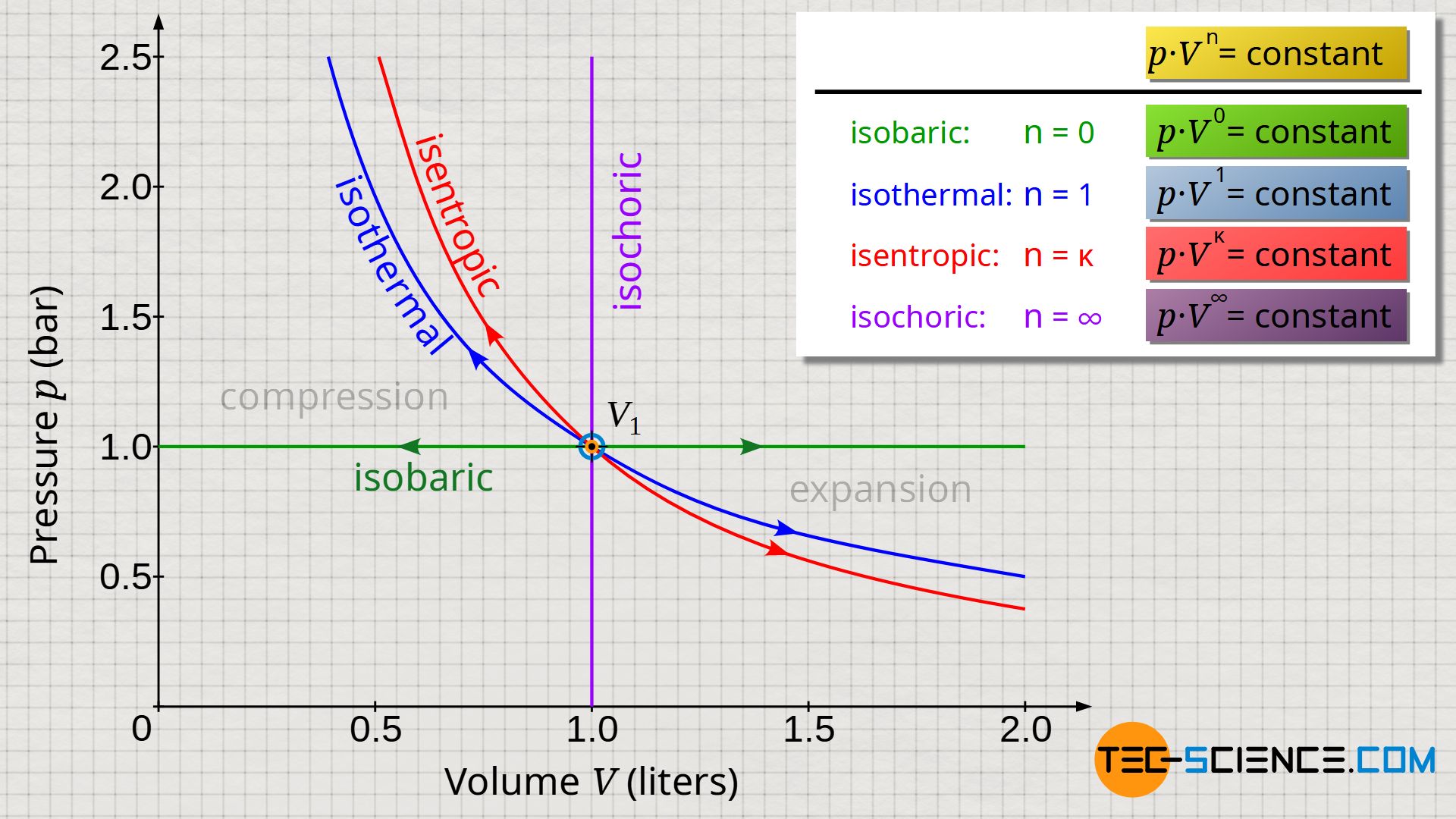
What is difference between isentropic and polytropic process? Then the value of n for which molar heat capacity of the process is negative is given as. If gamma is the ratio of specific heat CP/CV. What is an ideal polytropic gas?Ī polytropic process for an ideal gas is represented by the equation PVn constant. … If the energy of a system changes because of a temperature difference, we say there has been a flow of heat. The key difference between adiabatic and polytropic processes is that in adiabatic processes no heat transfer occurs whereas in polytropic processes heat transfer occurs. What is the difference between adiabatic and polytropic process? the case n =, pV = constant, corresponds to an isentropic (constant-entropy) process.the case n = 1, pV = constant, corresponds to an isothermal (constant-temperature) process.the case n = 0, p= constant, corresponds to an isobaric (constant-pressure) process.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
